Publications
AFNOR Certification certifies that the management system implemented by: NEWSOM for the manufacturing and distribution of mandibular advancement device has been assessed and found to meet the requirements. Certificat ISO 13485 : 2016 Read more
Predicting treatment response to mandibular advancement therapy using a titratable thermoplastic device Clinical Oral invesgations, 2021 – Trzepizur et al - Read more
Inclusion of BLUEPRO in the List of Reimbursable Products and Services (LPPR) Journal Officiel, 2019 - Read more
Comparison of titrable thermoplastic versus custom-made mandibular advancement device for the treatment of obstructive sleep apnoea Respiratory Medicine, 2017 - Gagnadoux et al - Read more
Pilot Study of a New Adjustable Thermoplastic Mandibular Advancement Device for the Management of Obstructive Sleep Apnoea-Hypopnoea Syndrome: A Brief Research Letter The Open Respiratory Medicine Journal, 2016 - El Ibrahimi et al - Read more
In vitro retention of a new thermoplastic titratable mandibular advancement device F1000 Research, 2015 - Braem - Read more
New solution for snoring is launched Dentistry, 2015 - Read more
Alleviate symptoms of problem snoring British Dental Journal, 2015 - Read more
Management of OSAHS by mandibular advancement orthosis in Algeria Poster, 2015 - Madoui et al - Read more

 |
|  |
|  |
|  |
| 





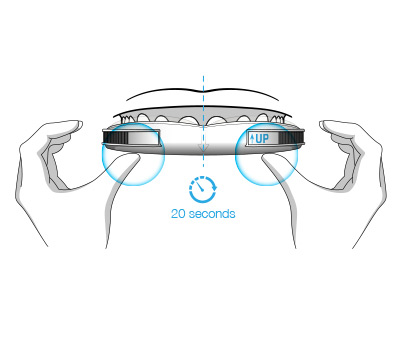
 The durable impression created in BluePro’s mentholated thermoplastic lining ensures a retentive force of 26 Newtons.This has been shown to be sufficient to resist jaw opening during sleep, ensuring the device remains effective throughout the night.
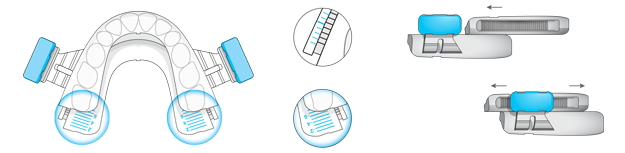
BluePro is a professional treatment option used exclusively under the supervision of health professionals. It provides an immediate, effective and affordable solution for the treatment of snoring and obstructive sleep apnea.
The durable impression created in BluePro’s mentholated thermoplastic lining ensures a retentive force of 26 Newtons.This has been shown to be sufficient to resist jaw opening during sleep, ensuring the device remains effective throughout the night.
BluePro is a professional treatment option used exclusively under the supervision of health professionals. It provides an immediate, effective and affordable solution for the treatment of snoring and obstructive sleep apnea.